WASHINGTON (AP) — The Food and Drug Administration said Friday it will add a new warning and other limitations to a gene therapy for Duchenne’s muscular dystrophy that's been linked to two patient deaths.
The infused therapy from Sarepta Therapeutics will carry a boxed warning — the most serious type — alerting doctors and patients to the risk of potentially fatal liver failure with the treatment, the FDA said in a release.
The one-time therapy, Elevidys, has been under FDA scrutiny since the company reported the first of two deaths of teenage boys in March. Following a second death reported in June, the FDA briefly called for halting all shipments of the drug. But the agency quickly reversed course after facing pushback from patient families and libertarian activists close to President Donald Trump.
Elevidys is the first U.S.-approved gene therapy for Duchenne’s muscular dystrophy, a fatal muscle-wasting disease that affects boys and young men.
In addition to the boxed warning, the FDA is also limiting the drug's approved use to patients who are 4 years old and up and can still walk. Previously the FDA had allowed the drug's use in immobile patients, who generally have more advanced disease.
New labeling will also recommend weekly liver function monitoring for the first three months of treatment, as well as other precautionary steps.
Elevidys is Sarepta's best-selling product and recent headwinds against the drug have weighed heavily on the company and its stock. In July, the Cambridge, Massachusetts-based company announced it would lay off 500 employees.
Sarepta Therapeutics Inc. shares rose 7.7% in trading after the FDA announcement, reflecting improved visibility for investors about the company's outlook.
___
The Associated Press Health and Science Department receives support from the Howard Hughes Medical Institute’s Department of Science Education and the Robert Wood Johnson Foundation. The AP is solely responsible for all content.
LATEST POSTS
- 1
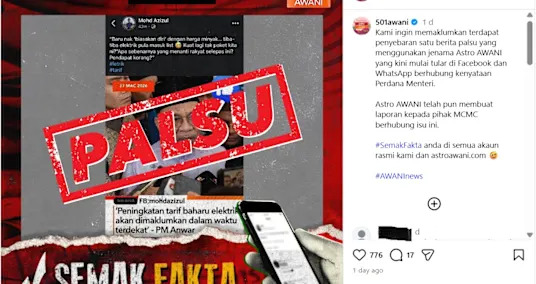
 Malaysian broadcaster rejects altered graphic about electricity rate hike
Malaysian broadcaster rejects altered graphic about electricity rate hike - 2
 Rights group: At least 2,500 deaths during protest crackdown in Iran
Rights group: At least 2,500 deaths during protest crackdown in Iran - 3
 Climate leaders are talking about 'overshoot' into warming danger zone. Here's what it means
Climate leaders are talking about 'overshoot' into warming danger zone. Here's what it means - 4
 Sheinelle Jones will cohost fourth hour of 'Today' with Jenna Bush Hager: Here's what to know about her
Sheinelle Jones will cohost fourth hour of 'Today' with Jenna Bush Hager: Here's what to know about her - 5
 19 Strange Motion pictures You Shouldn't Watch With Your Mum
19 Strange Motion pictures You Shouldn't Watch With Your Mum
 Scientists solve the mystery of the prehistoric 'Burtele Foot'
Scientists solve the mystery of the prehistoric 'Burtele Foot' 10 Moving Design Frill for Summer 2023
10 Moving Design Frill for Summer 2023 'Stranger Things' character guide: The nerds, the newcomers and the rest of the Season 5 cast
'Stranger Things' character guide: The nerds, the newcomers and the rest of the Season 5 cast Top 10 Arising Advances That Will Shape What's in store
Top 10 Arising Advances That Will Shape What's in store From a new flagship space telescope to lunar exploration, global cooperation – and competition – will make 2026 an exciting year for space
From a new flagship space telescope to lunar exploration, global cooperation – and competition – will make 2026 an exciting year for space The Fragrant Small Tree Birds & Pollinators Love With Stunning Flowers In Summer
The Fragrant Small Tree Birds & Pollinators Love With Stunning Flowers In Summer I binged all 24 Hallmark Christmas movies in less than 30 days. I emerged a changed man.
I binged all 24 Hallmark Christmas movies in less than 30 days. I emerged a changed man. Ukraine's new defense minister just outlined how dire its troop shortage has become
Ukraine's new defense minister just outlined how dire its troop shortage has become Nitty gritty Manual for Picking Agreeable Tennis shoes
Nitty gritty Manual for Picking Agreeable Tennis shoes